The pressure exerted by 6.0 g of methane gas in a 0.03 m^3 vessel at 129^∘C is :(Atomic masses : C = 12.01, H = 1.01 and R = 8.314 JK^-1 mol^-1)
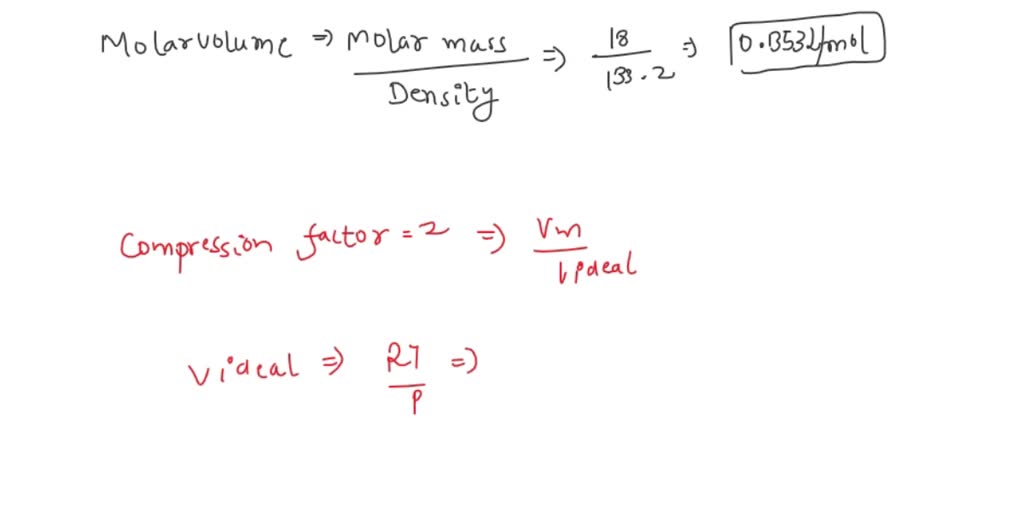
SOLVED: The mass density of water vapour at 327.6 atm and 776.4 K is 133.2 kg m−3. Given that for water a = 5.464 dm6 atm mol−2, b = 0.03049 dm3 mol−1,

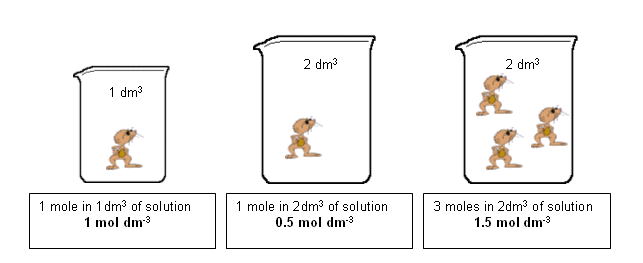
Calculating molarity units molar concentration of solutions practice questions on molarity how to make up a standard solution how to determine solubility gcse chemistry igcse KS4 science A level GCE AS A2

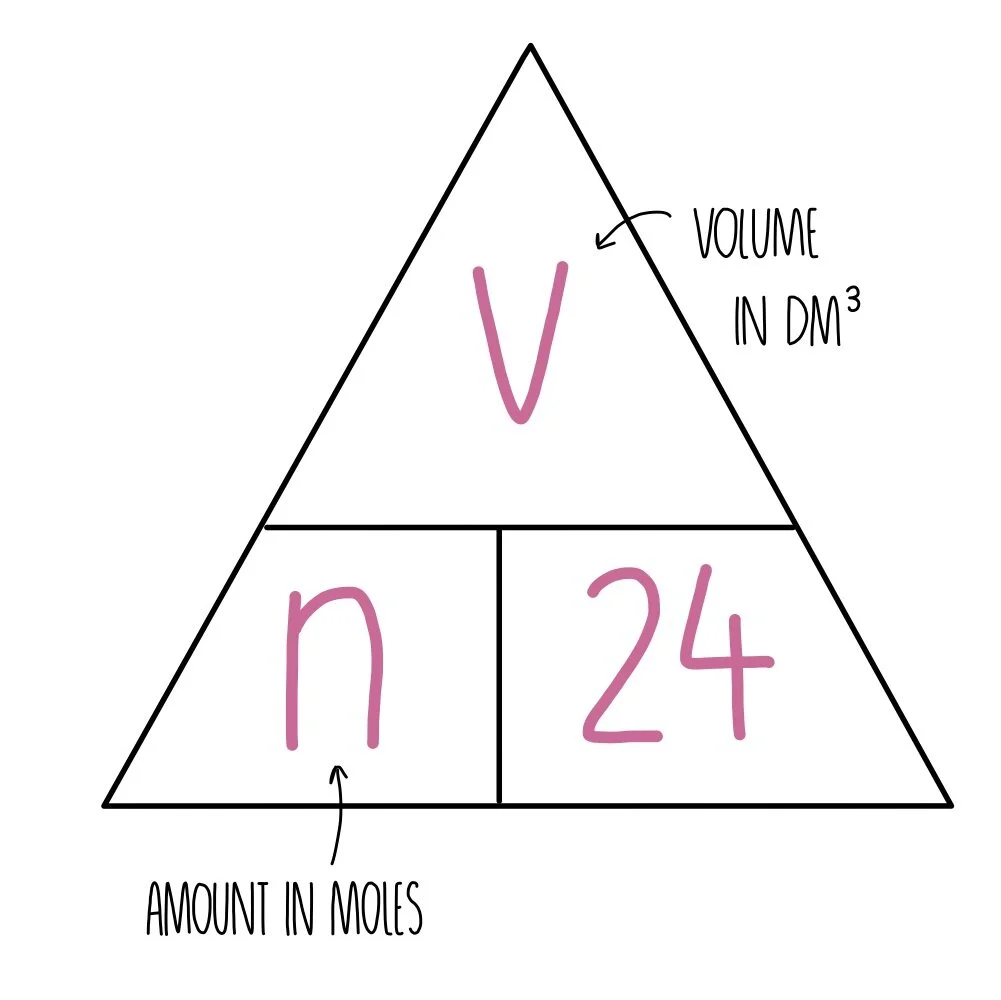
molar gas volume Avogadro's Law moles and mass calculations gcse chemistry calculations igcse KS4 science A level GCE AS A2 O Level practice questions exercises
Electronic absorbance spectra of 1.93 mmol dm −3 neptunium(IV) in 0.50... | Download Scientific Diagram

Basic chemical calculations. When solving numerical problems, always ask yourself whether your answer makes sense !!! - ppt download

The mass of 5.6 dm^3 of a certain gas at STP is 12.0 g . Calculate the relative molecular mass of the gas?
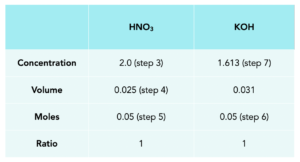
What volume of 0.1mol/dm3 hydrochloric acid will be required to neutralize 20cm3 of 2.0mol/DM3 sodium hydroxide? - Quora